ELN Software, the complete guide to Electronic Laboratory Notebook

Table of Contents
How to choose an ELN
The Complete Guide to Electronic Lab Notebook Software (2026)
Last updated: February 2026 | Reviewed by the LabSoftwareGuide editorial team
What this guide covers: What ELN software is and how it differs from paper notebooks, the five ELN types, core features to evaluate, an independent overview of 14 platforms organized by market segment, how to choose by lab type, compliance requirements, pricing guidance, and a practical buying guide. |
What Is an Electronic Lab Notebook (ELN)?
An Electronic Lab Notebook (ELN) — also called a digital lab notebook or electronic laboratory notebook — is software designed to replace the paper lab notebooks that scientists have used for centuries to record experiments, methods, observations, and results.
The global ELN market is experiencing strong growth, driven by the need for better data integrity, remote collaboration, regulatory compliance, and the progressive integration of AI into research workflows.
At its core, an ELN answers the questions that sit behind every scientific result:
What exactly was done, and by whom?
When was each step performed, and under what conditions?
What were the raw results, and have they been changed since recording?
Could another scientist reproduce this experiment from these records?
Is this data valid, traceable, and compliant with applicable regulations?
Paper notebooks cannot reliably answer most of these questions. ELN software can — provided you choose the right one for your specific environment.
The 5 Types of ELN Software: Which One Fits Your Lab?
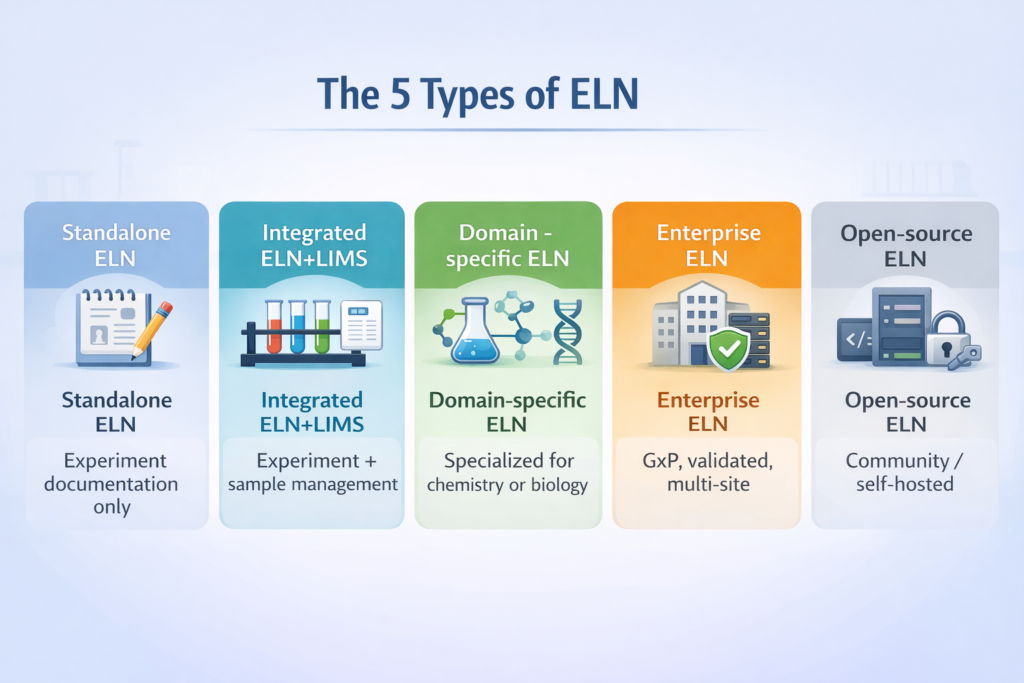
Not all ELN platforms are the same. Before comparing vendors, understanding the five distinct categories of ELN software is essential — choosing the wrong type is a bigger mistake than choosing the wrong vendor within the right type.
ELN Type | Description | Best For | Examples |
|---|---|---|---|
Standalone ELN | Pure digital notebook: experiment docs, protocols, data capture, search, audit trails | Academic labs, small research teams, document-first workflows | LabArchives, RSpace, labfolder, SciNote |
Integrated ELN/LIMS | Combines experiment documentation with sample tracking, inventory, and workflow management | Biotech, pharma R&D, labs needing both ELN and LIMS in one platform | Benchling, Labguru, eLabJournal, LabWare ELN |
Domain-specific ELN | Specialized tools for chemistry (structure drawing, reactions) or biology (sequences, plasmids) | Medicinal chemistry, genomics, antibody development | Dotmatics, Revvity Signals, Arxspan |
Enterprise ELN | Scalable, multi-site, GxP-validated, IT governance, single sign-on, full compliance audit trail | Large pharma, CROs, enterprise orgs with strict compliance requirements | IDBS E-WorkBook, Revvity Signals, Thermo Fisher Core ELN, Dotmatics |
AI-native ELN | 3rd-gen platforms using AI/ML to assist protocol drafting, data extraction, and experiment analysis | Cutting-edge biotech and labs accelerating with AI-assisted research | Benchling (AI features), Labstep |
Open-source ELN | Community or self-hosted ELN with full source code and no licensing fees | Academic institutions, budget-constrained or privacy-focused labs | eLabFTW, OpenBIS, RSpace (OSS) |
Key insight: The most common mistake labs make is buying a standalone ELN when they need an integrated ELN/LIMS — or overspending on an enterprise platform when a simpler academic tool would serve their current stage of growth. |
14 ELN Platforms: Independent Market Overview (2026)
The table below provides an independent overview of 14 widely used ELN platforms, organized by market segment. No vendor has paid for inclusion or positioning. Pricing is intentionally not included — published rates vary significantly by configuration, contract size, and region. Contact vendors directly for current figures.
ELN Platform | Vendor / Parent | Category | Deployment | Best For |
|---|---|---|---|---|
ENTERPRISE LEADERS — pharma, biotech, regulated environments, large-scale deployments | ||||
Dotmatics ELN | Dotmatics (Insightful Science) | Enterprise ELN + R&D suite | Cloud + On-premise | Pharma, biotech, chemistry & materials R&D |
IDBS E-WorkBook | IDBS (Danaher Life Sciences) | Enterprise ELN | Cloud + On-premise | Enterprise pharma R&D, CROs, regulated workflows |
Revvity Signals Notebook | Revvity (ex-PerkinElmer) | Cloud-native enterprise ELN | Cloud SaaS | Pharma, biotech, chemistry & materials science |
Thermo Fisher Core ELN | Thermo Fisher Scientific | Enterprise ELN | Cloud | Enterprise labs; integrates with Thermo Core LIMS |
LabWare ELN | LabWare | ELN + LIMS integration | Cloud + On-premise | QC labs, process-driven and regulated environments |
CLOUD R&D REFERENCES — biotech teams that iterate fast | ||||
Benchling Notebook | Benchling | ELN + R&D cloud platform | Cloud SaaS | Biotech R&D (US focus), molecular biology teams |
Labguru ELN | Thermo Fisher (distributed) | ELN + inventory + workflows | Cloud SaaS | Biotech and academic-to-biotech R&D teams |
ACADEMIC & MID-MARKET — universities, institutes, SMEs | ||||
LabArchives | LabArchives | Standalone ELN | Cloud SaaS | Academic research, US universities, education |
SciNote | SciNote | ELN + inventory + compliance | Cloud SaaS | Academia, pharma, multi-country institutions |
RSpace | ResearchSpace | ELN + FAIR / open science workflows | Cloud + Self-hosted | Universities, institutes, open science communities |
labfolder | SciSure | Standalone ELN | Cloud SaaS | Academic and industrial documentation workflows |
eLabJournal | eLabNext | Connected lab ELN | Cloud SaaS | Research labs, modular connected lab operations |
Labstep | STARLIMS / Abbott | ELN + interactive protocols | Cloud SaaS | Protocol-driven labs, IP documentation, compliance traceability |
OPEN-SOURCE | ||||
eLabFTW | Open-source community | Open-source ELN | Self-hosted | Academic labs and tech-savvy orgs valuing control & privacy |
Note on deployment: Most modern ELN platforms offer cloud SaaS as their primary option. On-premise and hybrid deployment are typically available on enterprise plans or on request. Always confirm your data sovereignty and IT security requirements before selecting a deployment model. |
Core Features of ELN Software: What to Look For
Across all ELN types, the following capabilities define a modern, fit-for-purpose platform. Use this as your evaluation checklist.
1. Experiment Documentation and Structured Data Capture
Rich-text editor with support for images, files, tables, chemical structures, and sequences
Flexible entry templates and protocol libraries
Structured and unstructured data capture in a single workspace
Mobile capture support (tablet, smartphone) for at-bench recording
Offline mode for labs with restricted internet access
2. Search, Retrieval, and Knowledge Management
Full-text search across all entries, attachments, and metadata
Tagging, categorization, and cross-referencing between experiments
Protocol versioning and reuse across multiple experiments
Project and group-level organization hierarchies
3. Collaboration and Access Controls
Real-time multi-user editing and commenting
Role-based access control (scientist, reviewer, PI, admin)
Cross-site collaboration with granular permission settings
Shared protocol libraries accessible across teams or departments
4. Audit Trails, Electronic Signatures, and Data Integrity
Immutable, timestamped audit trail for all entries and edits
21 CFR Part 11-compliant electronic signatures
Witness signature workflows
Data locking with controlled amendment processes
Activity logs: who accessed what, and when
5. Integration with Laboratory Instruments and Systems
Instrument data import (direct interface or file import)
Integration with LIMS for sample-experiment linkage
SDMS connectivity for raw data file management
API availability for custom integrations
6. Compliance and Regulatory Support
21 CFR Part 11 (FDA, USA) — electronic records and signatures
EU GMP Annex 11 — computerized systems in GMP environments
GLP (Good Laboratory Practice) compliance
ISO 17025 — testing and calibration labs
Institutional IP protection and legal witness workflows
7. AI and Automation Capabilities
Leading platforms are beginning to offer AI-assisted capabilities that go beyond search:
AI-assisted protocol drafting and experiment design suggestions
Automated data extraction from instrument outputs
Natural language search and query across all lab data
Pattern identification and anomaly detection across experimental datasets
Evaluator’s tip: Ask vendors for specific, documented examples of AI use cases — not marketing claims. Request a live demo of the AI feature applied to a workflow similar to yours before factoring it into your decision. |
ELN vs Paper Notebooks vs Word/Excel: A Practical Comparison

Many labs still debate whether the investment in ELN software is justified. Here’s an objective breakdown of what each approach delivers:
Dimension | Paper Lab Notebook | Word / Excel | ELN Software |
|---|---|---|---|
Searchability | Manual index only | Limited text search | Full-text, instant search |
Audit trail | None | None | Timestamped, immutable |
Collaboration | Single user / photocopy | Version conflicts | Real-time multi-user |
IP protection | Witness signatures only | Weak / uncontrolled | Electronic signature + timestamp |
Compliance | Manual SOPs | Not fit for regulated labs | Built-in compliance tooling |
Scalability | Shelf space limits | Performance issues at scale | Cloud-native, unlimited |
Data integrity | Risk of loss or damage | Editable without trace | Locked, versioned records |
Integration | None | Manual export / import | Instrument + LIMS + SDMS APIs |
The tipping point for most labs comes when one of three things happens: a compliance audit reveals inadequate record-keeping, a key researcher leaves and takes institutional knowledge with them, or a failed reproducibility attempt exposes gaps in experimental documentation.
ELN Software by Lab Type and Industry
The right ELN is rarely universal. Requirements differ substantially by scientific discipline, regulatory context, organizational size, and research stage.
Academic Research Laboratories
Academic labs balance tight budgets against the need for a tool scientists will actually adopt. Complexity is the enemy of uptake.
Free or low-cost entry tier is essential for labs without dedicated software budgets
Flexible structure that accommodates diverse research types (chemistry, biology, physics)
Integration with PubMed, protocols.io, and institutional repositories
Easy multi-PI and multi-group access management
Data export in open formats to avoid vendor lock-in
Platforms commonly chosen: LabArchives, SciNote, RSpace, labfolder
Pharmaceutical R&D and Drug Discovery
Pharma R&D labs operate under some of the tightest regulatory constraints in the world. An ELN is a regulatory document as much as a scientific tool.
Full 21 CFR Part 11 compliance with validated electronic signatures
GxP-aligned workflows and SOPs with deviation management
Integration with LIMS for sample and compound linkage
Chemistry-specific tools: structure drawing, reaction management, compound registration
Vendor-supplied IQ/OQ/PQ validation packages
Full data integrity controls meeting ALCOA+ principles
Platforms commonly chosen: Dotmatics, IDBS E-WorkBook, Revvity Signals Notebook, Benchling, Thermo Fisher Core ELN
Biotech Startups and Scale-ups
Biotech startups need an ELN that can grow with them — from a founding team to a Series B organization with regulatory submissions on the horizon.
Fast onboarding with minimal IT overhead
Scalable architecture that does not require renegotiation at each growth stage
21 CFR Part 11 capability ready to activate when IND-enabling studies begin
Strong molecular biology tooling: sequence editor, clone management, plasmid maps
Platforms commonly chosen: Benchling, Labguru, SciNote, eLabJournal
Contract Research and Manufacturing Organizations (CROs / CMOs)
CROs and CMOs operate across multiple client projects simultaneously, with strict IP segregation and rigorous audit trail requirements from every sponsor.
Project-level access controls enforcing client data segregation
Sponsor-level audit trail access with configurable permissions
Study management across multiple simultaneous client engagements
Regulatory submission-ready record exports
Platforms commonly chosen: IDBS E-WorkBook, Dotmatics, Revvity Signals Notebook, Benchling
QC and Regulated Manufacturing Laboratories
QC labs need ELNs built around structured, procedure-driven documentation with high reliability and tight process control.
Template-driven entry for standardized test methods and procedures
SOP execution workflows with step-by-step compliance tracking
Integration with LIMS for result and CoA generation
Strong audit trail and electronic signature for batch record support
Platforms commonly chosen: LabWare ELN, Thermo Fisher Core ELN, Revvity Signals Notebook
Academic-to-Industry Transition Labs
Research teams starting in academic settings that need to build compliance capabilities as they mature toward commercialization.
Accessible entry pricing that doesn’t create a barrier at the founding stage
Clear upgrade path to compliance-grade features as regulatory needs emerge
Active user communities sharing protocols and best practices
Platforms commonly chosen: Labguru, SciNote, eLabJournal, Labstep, RSpace
ELN Platform Reviews: Independent Editorial Assessment
The following reviews reflect independent editorial assessment based on available product documentation, vendor positioning, and market knowledge. No vendor has paid for inclusion, positioning, or specific language in these reviews.
Enterprise Leaders
Dotmatics ELN

Dotmatics has built one of the most comprehensive scientific informatics ecosystems in the market, following a series of acquisitions that brought together tools including GraphPad Prism, Geneious, and others under a unified platform strategy. The ELN module sits at the centre of that ecosystem, with particularly strong capabilities for chemistry and multi-omics R&D: chemical structure handling, reaction management, and compound registration are deeply integrated into the documentation workflow. The platform is genuinely designed for organizations working across multiple scientific disciplines simultaneously. The practical consideration: Dotmatics is best evaluated as part of a broader informatics strategy. Organizations without complex multi-disciplinary requirements may find the entry investment disproportionate to their needs.
Best for: Pharmaceutical and chemistry R&D organizations, multi-disciplinary research teams, organizations already in or evaluating the broader Dotmatics suite.
IDBS E-WorkBook

IDBS E-WorkBook is a mature, enterprise-grade ELN with a long track record in pharmaceutical, CRO, and life science R&D. Now part of Danaher Life Sciences, it positions itself as a core component of the broader Danaher informatics portfolio. Its depth in regulatory compliance is well-established: the platform has been deployed in environments that have undergone FDA inspection and GxP audit. For evaluators, the realistic consideration is implementation complexity and timeline — E-WorkBook deployments in regulated environments are typically measured in months and require significant internal resource for validation activities.
Best for: Large pharmaceutical companies, global CROs, enterprise R&D organizations with significant regulatory compliance requirements.
Revvity Signals Notebook

Following the split of PerkinElmer into Revvity and PerkinElmer Instruments, Signals Notebook sits within the Revvity informatics portfolio. It is a cloud-native enterprise ELN with established presence across pharmaceutical, biotech, chemistry, and materials science environments. The platform’s strengths include collaborative data capture, handling of scientific data across modalities, and integration with other Revvity informatics tools. User feedback has noted that the interface can feel complex for scientists new to enterprise ELN platforms, and that configuration depth — while powerful — requires dedicated administrator effort.
Best for: Enterprise pharma, biotech, and chemistry / materials science organizations requiring a cloud-native, compliance-ready ELN platform.
Thermo Fisher Core ELN

Thermo Fisher’s Core ELN is the ELN component of the broader Core informatics platform, designed to work in close integration with Core LIMS. For organizations already within the Thermo Fisher ecosystem — or those evaluating a combined ELN/LIMS implementation with a single vendor — this integration is a genuine practical advantage. The platform targets enterprise-scale research environments and regulated labs. Organizations evaluating it independently of the broader Thermo Fisher platform relationship should confirm that standalone deployment meets their specific workflow requirements.
Best for: Enterprise labs already in or evaluating the Thermo Fisher informatics ecosystem; organizations seeking tight ELN/LIMS integration under a single vendor.
LabWare ELN

LabWare is best known as a leading LIMS vendor, and its ELN offering is most compelling in the context of that broader LabWare platform relationship. The ELN module is designed to complement the LIMS in procedure-driven, QC-heavy environments — capturing the experimental record alongside the sample management and workflow functions that LabWare LIMS handles. For organizations already running LabWare LIMS, the ELN extension is a natural consolidation. For organizations seeking a standalone ELN, the evaluation should focus on whether the workflow model fits their specific documentation needs.
Best for: QC and regulated manufacturing labs already using LabWare LIMS; process-driven environments where ELN and LIMS integration is the primary requirement.
Cloud R&D References
Benchling Notebook

Benchling has become the de facto standard ELN for well-funded US biotech R&D, and its molecular biology tooling remains best-in-class: sequence editor, plasmid map viewer, primer management, and entity registry are deeply embedded in the experiment workflow. The platform has expanded well beyond a simple ELN — Benchling’s R&D cloud covers entity registration, study management, and process modelling, positioning it as a central informatics hub for the discovery lab. Practical considerations for evaluators: Benchling’s pricing is structured for funded biotech, data migration out of the platform requires planning, and the feature depth can be disproportionate for organizations with straightforward documentation needs.
Best for: Biotech and life science R&D organizations, molecular biology teams, companies scaling from early discovery toward IND-enabling work.
Labguru ELN

Labguru, distributed by Thermo Fisher Scientific, positions itself as an all-in-one platform covering ELN, LIMS, and inventory management. It has a solid track record with biotech and academic-to-biotech research teams that want to consolidate multiple functions under a single platform without the complexity and cost of enterprise-grade solutions. The platform includes molecular biology and chemistry tools, workflow management, and collaboration features suited to growing research teams. Teams with highly complex or specialized workflows may find that some configurations benefit from vendor support to implement effectively.
Best for: Biotech R&D teams, academic-to-biotech organizations, labs seeking an all-in-one ELN/LIMS platform without enterprise-level complexity.
Academic and Mid-Market Platforms
LabArchives

LabArchives is one of the most widely adopted ELN platforms in US academic institutions, with a strong presence in research universities and educational settings. Its pricing model — accessible for academic use — has driven high uptake, and its simplicity and reliability have made it a trusted first ELN for researchers transitioning from paper. It integrates with a range of academic platforms and LMS tools. The appropriate audience is academic and educational labs prioritizing straightforward, dependable experiment documentation over advanced compliance or automation features.
Best for: US research universities, educational laboratories, academic collaborations, individual researchers in academic settings.
SciNote

SciNote has built a strong reputation for balancing usability with genuine compliance capability. It is one of the few ELN platforms with credible adoption across both FDA/USDA-regulated environments and small academic labs. The free plan provides real functionality for individual researchers, and paid tiers progressively add inventory management, team collaboration, and compliance features. Its communication around multi-country adoption and institution-level case studies is among the most transparent in the market. The ceiling on platform complexity is real: labs with highly specialized scientific workflows may eventually require more domain-specific tooling.
Best for: Academic labs, research institutions, teams making their first transition from paper notebooks, small-to-mid-sized organizations with compliance requirements.
RSpace (ResearchSpace)

RSpace takes a distinctive position in the ELN market with its emphasis on FAIR data principles, open science, and interoperability. It is widely adopted in European and US research institutes and universities, and its approach to data provenance and external repository integration sets it apart from more workflow-centric platforms. RSpace is available both as a cloud service and as a self-hosted open-source version, giving institutions flexibility on data sovereignty. It is a strong choice for academic environments where open science commitments and long-term data accessibility are priorities.
Best for: Research universities, public research institutes, academic groups with open science or FAIR data commitments, organizations wanting cloud or self-hosted flexibility.
labfolder (SciSure)

labfolder, now part of SciSure, is one of the most accessible ELN options on the market — fast to deploy, easy to learn, and designed to minimize friction for scientists transitioning from paper. It handles structured documentation reliably and supports both academic and industrial use cases. The platform is particularly appreciated for its clean documentation experience and straightforward onboarding. Labs with specialized scientific workflows or demanding compliance validation requirements will find the platform’s ceiling on complexity relatively low.
Best for: Small academic and industrial labs, individual researchers, organizations prioritizing ease of adoption and documentation clarity over platform breadth.
eLabJournal (eLabNext)

eLabJournal is the ELN component of the eLabNext connected lab platform, which also includes eLabInventory and eLabProtocols. The modular approach is a genuine advantage: labs can start with ELN alone and layer in inventory and protocol management as needs evolve, without migrating to a different vendor. The platform handles core ELN workflows reliably, and the eLabNext ecosystem provides a credible growth path for labs that anticipate broader connected lab needs over time.
Best for: Academic and biotech labs wanting a modular platform that can grow from ELN-only into a broader connected lab system.
Labstep (STARLIMS)

Labstep, now part of the STARLIMS portfolio (Abbott), is designed around interactive protocol execution and real-time data capture at the bench. Its design philosophy — making protocol steps traceable and interactive — makes it well-suited to labs where IP documentation, reproducibility, and compliance traceability are central concerns. The STARLIMS association brings enterprise credibility and integration potential, while Labstep’s own interface remains modern and accessible. It is worth evaluating for organizations that find traditional ELN interfaces too unstructured for their step-by-step protocol compliance requirements.
Best for: Protocol-driven research labs, organizations prioritizing IP documentation and compliance traceability, labs evaluating structured protocol execution over free-text documentation.
Open-Source
eLabFTW

eLabFTW is the most widely adopted open-source ELN, with an active development community and a self-hosted deployment model that gives organizations full control over their data and infrastructure. It covers core ELN functions — experiment documentation, sample and object management, protocol management, and team collaboration — with no licensing fees. It is genuinely capable for academic and research environments with the technical capability to install and manage a self-hosted application. The critical gap for regulated labs is the absence of vendor-supplied validation documentation: organizations subject to 21 CFR Part 11 or similar requirements would need to manage their own validation effort internally.
Best for: Academic institutions with IT capability to self-host, organizations with data sovereignty requirements, budget-constrained labs comfortable with open-source infrastructure.
How to Choose an ELN: The Lab Manager’s Buying Guide
With over 50 ELN platforms on the market, the challenge is filtering them intelligently. The following six-step framework helps lab managers make defensible, long-term decisions.
Step 1: Clarify Your Scientific Workflow First
Document what your lab actually needs to record before looking at any platform.
Chemistry labs need structure drawing, reaction tracking, and spectral data attachment
Molecular biology labs need sequence editors, primer databases, and plasmid management
In vivo labs need study management with regulatory traceability
Analytical and QC labs need method execution templates and result validation workflows
An ELN that cannot natively handle your data types will generate workarounds that erode adoption over time.
Step 2: Identify Non-Negotiable Compliance Requirements
Compliance requirements are binary. Map these before any vendor conversation and treat them as explicit disqualifiers. Here your can find our specific article about ELN Compliance and data integrity.
FDA regulated: 21 CFR Part 11
EU pharmaceutical: Annex 11 / EMA GMP
Research labs: GLP where applicable
Testing and calibration: ISO 17025
Step 3: Decide on Deployment Model Early
Cloud / SaaS: Preferred by most labs for accessibility, automatic updates, lower IT overhead
On-premise: Required by some government labs, defense organizations, and certain regulated environments
Private cloud / hybrid: Increasingly available from enterprise vendors
Step 4: Calculate True Total Cost of Ownership (TCO)
License fees (per-user vs flat-rate vs module-based)
Implementation and onboarding fees
Validation costs (IQ/OQ/PQ) for regulated labs
Training costs for initial deployment and ongoing staff turnover
Annual maintenance, support, and upgrade fees
Internal IT time for administration and integrations
Cost reality check: A ‘free’ open-source ELN is not free. Self-hosting, configuration, security patching, user support, and validation can cost more in staff time than a mid-market commercial subscription. Always model the full picture before committing. |
Step 5: Run a Realistic Pilot with Actual Users
A 30-day pilot with the scientists who will use the system every day — using real experiments from your own workflows — will reveal usability issues, missing features, and integration gaps that no vendor demo will surface.
Step 6: Evaluate the Vendor Relationship, Not Just the Software
How is the platform funded? What happens if they’re acquired?
What do their SLA response times and support tiers look like?
What do current customers say about validation support?
What does data portability look like if you need to migrate?
Open-Source ELN Software: When It Makes Sense
Open-source ELN platforms offer full source code access, no licensing fees, and the freedom to customize without vendor permission. The most widely used option is eLabFTW, with OpenBIS and the self-hosted RSpace also present in institutional settings.
Open-source ELN is a good choice for:
Academic institutions with dedicated IT staff and limited software budgets
Labs with unique workflow requirements commercial platforms cannot configure
Organizations with strong data privacy requirements and technical capability to self-host
Open-source ELN is a poor choice for:
Labs without dedicated IT or developer resources
Regulated labs that need vendor-supplied validation documentation
Organizations expecting commercial-level support SLAs and feature roadmap commitment
Important: Open-source does not mean free. Hosting infrastructure, security maintenance, validation effort, and internal IT support can easily exceed the cost of a mid-market commercial subscription. Run a realistic TCO comparison before committing. |
ELN Market Trends in 2026
1. AI Integration Is Moving from Hype to Tested Utility
AI-assisted protocol drafting, automated data extraction from instruments, and natural language search across experimental archives are now available in several platforms. The practical challenge is validating AI output quality against real scientific workflows — marketing claims need to be tested in live demos before factoring AI into a purchasing decision.
2. Convergence of ELN, LIMS, and SDMS
The boundary between ELN, LIMS, and SDMS is gradually dissolving. Enterprise vendors are converging on unified informatics platforms that treat experiment documentation, sample management, and raw data storage as layers of a single system. For labs evaluating new ELN deployments, the question is increasingly ‘What is our informatics strategy for the next decade?’ rather than ‘Which ELN?’
3. FAIR Data Principles Are Becoming Standard Requirements
Funding bodies (NIH, ERC), regulatory agencies, and pharma sponsors are increasingly requiring FAIR data compliance (Findable, Accessible, Interoperable, Reusable). ELN platforms that can demonstrate FAIR-aligned data architecture have a meaningful advantage in regulated and grant-funded environments.
4. Cloud-First Is Now the Default, Even in Regulated Environments
Cloud-native ELN deployments are now standard even in heavily regulated pharma environments. Major vendors offer validated SaaS options designed to satisfy 21 CFR Part 11 and GMP requirements. The question has shifted to which cloud architecture best fits your organization’s security and governance requirements.
5. Free and Freemium ELNs Are Raising the Academic Adoption Floor
Genuinely capable free ELN tiers have effectively eliminated the cost barrier to ELN adoption in academic settings, creating a generation of early-career scientists who enter industry already accustomed to ELN workflows — accelerating commercial adoption timelines.
ELN Software Pricing: What to Expect
ELN pricing spans an unusually wide range — from genuinely free to large annual enterprise contracts. The table below provides segment-level guidance. Because published rates vary significantly between vendors, by configuration, and by region, vendor-specific pricing is not included. Contact vendors directly for current figures.
Segment | Price Range | Typical Users |
|---|---|---|
Free / freemium | Free (limited features or storage) | Academic researchers, students, individual scientists — free tiers exist on several platforms across the market |
SME / startup | From a few dollars to ~$100–200/user/month | Biotech startups, small CROs, university spin-offs — budget for team plans varies significantly by platform and region |
Mid-market | Often $200–$800+/user/month or flat annual fee | Mid-sized pharma R&D, diagnostics labs, growing biotech teams needing compliance features and broader integration |
Enterprise | Custom quote — typically $50K to $500K+/year | Large pharma, global CROs, hospital networks — enterprise contracts include implementation, validation, and support packages |
Hidden cost warning: Enterprise ELN pricing typically excludes implementation services, validation packages, training, and ongoing customization — which can match or exceed the annual license cost. Always request a total first-year cost estimate including all professional services before comparing platforms on license cost alone. |
Frequently Asked Questions About ELN Software
What does ELN stand for?
ELN stands for Electronic Lab Notebook (or Electronic Laboratory Notebook). It is software that replaces paper lab notebooks, providing a digital environment for recording experiments, protocols, observations, and results.
What is the difference between an ELN and a digital lab notebook?
The terms are used interchangeably. ‘Electronic lab notebook’, ‘electronic laboratory notebook’, ‘digital lab notebook’, and ‘ELN’ all refer to the same category of software. Some vendors prefer ‘digital lab notebook’ in their marketing to appeal to audiences less familiar with the ELN acronym.
What is the difference between an ELN and a LIMS?
An ELN is primarily focused on research documentation: capturing what was done, why, and how. A LIMS (Laboratory Information Management System) is focused on operational management: tracking samples, workflows, test results, and compliance. Many modern platforms now combine both functions. If your primary need is experiment documentation, start with ELN. If you primarily need sample tracking and QC workflow management, start with LIMS.
Is ELN software required for regulatory compliance?
ELN software is not universally required by regulation, but it is increasingly the expected standard for labs operating under 21 CFR Part 11 (FDA), EU GMP Annex 11, GLP, or ISO 17025. Paper notebooks can technically satisfy regulatory requirements, but the practical difficulty of meeting audit trail and electronic signature expectations makes ELN adoption the de facto compliance path for most regulated labs.
Can a free ELN be used in a regulated environment?
It depends on the platform and the specific tier. Some free ELN tiers include basic audit trail features. However, 21 CFR Part 11 compliance typically requires electronic signature capabilities and validation documentation — usually available only on paid plans. For regulated labs, confirm explicitly which compliance features are included in the tier you are evaluating before making any assumptions.
How long does it take to implement an ELN?
For standalone cloud ELNs with small teams: days to weeks. For mid-market platforms with custom configuration: 1–3 months. For enterprise ELN implementations with full GxP validation (IQ/OQ/PQ): 6–18 months. The biggest variable is validation: regulated labs should budget significantly more time and resource for validation activities than for the technical implementation itself.
What is 21 CFR Part 11 compliance in the context of ELN?
21 CFR Part 11 is a US FDA regulation governing electronic records and electronic signatures in FDA-regulated environments. An ELN claiming 21 CFR Part 11 compliance must provide secure access controls, immutable audit trails, electronic signature capabilities with identity verification, and system validation documentation. If your lab submits data to the FDA or operates under FDA oversight, any ELN generating that data must meet these requirements.
How do I migrate from paper notebooks to an ELN?
A structured migration has four phases: (1) select and configure the ELN platform, (2) digitize or archive historical paper records, (3) train users and run a controlled pilot with new experiments, (4) formally retire paper notebooks with a recorded cutover date. The most critical success factor is user adoption — invest in training and change management at least as much as in the technical implementation.
What is the difference between an open-source and a commercial ELN?
Open-source ELNs offer free access to the source code and can be self-hosted and customized without licensing fees. Commercial ELNs provide vendor support, regular updates, validation packages, and implementation services at a cost. For regulated labs, the absence of vendor-supplied validation documentation in most open-source ELNs is a significant practical gap that must be managed internally.
Conclusion: Choosing the Right ELN for Your Laboratory
The ELN market in 2026 is mature, competitive, and evolving rapidly across all segments — from free academic tools to enterprise platforms serving as central informatics hubs for global pharmaceutical organizations.
The right platform depends more on your specific scientific workflow, compliance requirements, and organizational scale than on any universal ranking. For academic labs making their first transition from paper: LabArchives, SciNote, RSpace, or labfolder offer strong entry points with minimal friction. For biotech startups scaling toward regulatory submissions: Benchling or Labguru provide the right combination of scientific depth and compliance readiness. For QC and regulated manufacturing: LabWare ELN and Thermo Fisher Core ELN are built around the process-driven workflows these environments require. For large pharma and CROs under GxP: Dotmatics, IDBS E-WorkBook, and Revvity Signals Notebook offer the enterprise depth and validation support that regulated environments demand.
The most important advice: define your requirements before talking to vendors. Involve bench-level scientists in the evaluation. Run a real pilot with your own experiments. And evaluate the vendor relationship — not just the software — because you will be working with them for years.
This guide is updated regularly as the ELN market evolves. Bookmark labsoftwareguide.com for the latest independent coverage of laboratory software. |
Explore More ELN Resources on LabSoftwareGuide
Open-Source ELN Software: Pros and Cons — Critical Factors Every Lab Should Evaluate
ELN vs LIMS: Which Does Your Lab Actually Need?
Best ELN Software for Small Laboratories
Best ELN for Pharma and Regulated Environments
How to Write an ELN Requirements Document
ELN Implementation Guide: From Paper to Digital in 90 Days
21 CFR Part 11 Compliance for Electronic Lab Notebooks
Free ELN Software: Honest Review of the Best No-Cost Options
LabSoftwareGuide is an independent editorial resource. We do not accept payment for vendor rankings or editorial positions in our pillar guides. Some individual articles may contain clearly disclosed sponsored content or affiliate links. All reviews reflect independent research and the collective expertise of our editorial team.
Interested in getting in touch with our ELN partners?
About Lab Software Guide Editorial Team
Who are we ?
The LabSoftwareGuide editorial team is led by a laboratory informatics specialist with extensive experience in the commercial side of lab software — including LIMS, ELN, and bioinformatics platforms across pharma, biotech, and life science environments.
Our editorial approach is built on that experience: we understand how lab software is evaluated, purchased, and implemented, and we know the questions that lab managers, scientists, and IT teams actually struggle with when choosing between platforms.
How we work ?
Every guide and review on LabSoftwareGuide is produced through a combination of hands-on platform testing, vendor interviews, and analysis of real user feedback from G2, Capterra, and laboratory community forums. We cross-reference commercial claims against documented user experience to give you assessments that reflect reality — not marketing materials.
Our Editorial Independence
LabSoftwareGuide accepts sponsored content and commercial partnerships with software vendors. These are always clearly labeled. Our editorial guides, pillar pages, and independent reviews are produced separately from any commercial relationship and are not influenced by vendor partnerships.
Our focus: We cover laboratory information management systems (LIMS), electronic lab notebooks (ELN), and related lab software for professionals working in pharma, biotech, clinical, environmental, and research laboratories.

What is ALCOA+? Data Integrity in Laboratory Environments
This article is based on official regulatory sources: FDA’s 2018 Data Integrity and CGMP guidance, PIC/S PI 041, EMA’s 2010 Reflection Paper, MHRA’s GxP Data Integrity guidance, and WHO TRS

What is 21 CFR Part 11?
A Practical Guide for Lab Software in 2026 This guide is based on the official 21 CFR Part 11 regulatory text (eCFR), FDA’s Scope and Application Guidance (2003), FDA’s final

Open-Source ELN Software: Pros and Cons – Critical Factors Every Lab Should Evaluate
As laboratories accelerate digital transformation, many organizations explore alternatives to expensive proprietary platforms. One option that often attracts attention is Open-Source ELN Software: Pros and Cons. At first glance, open-source
