LIMS Software, the complete guide to laboratory information management Systems

Table of Contents
Choosing a LIMS Software
The Complete Guide to Laboratory Information Management Systems (2026)
Last updated: February 2026 | Reviewed by the LabSoftwareGuide editorial team
What this guide covers: What LIMS software is, how it works, the key features to look for, how to choose between vendors, industry-specific requirements, compliance considerations, and an independent comparison of the top LIMS platforms available today. |
What Is a LIMS? (Laboratory Information Management System)
A Laboratory Information Management System (LIMS) is software built to manage the operational core of a laboratory: samples, workflows, test results, data integrity, and regulatory compliance.
The concept dates back to the 1980s when large pharmaceutical and petrochemical labs first needed a digital alternative to paper-based tracking. Today, LIMS has evolved into a mission-critical platform used across pharma, biotech, clinical diagnostics, environmental testing, food and beverage, and academic research.
At its most basic, a LIMS answers three questions that every lab manager asks daily:
Where is this sample right now, and what happened to it?
Are my results accurate, complete, and traceable?
Am I compliant with the applicable regulations and standards?
Modern LIMS platforms go well beyond basic sample tracking. They integrate with laboratory instruments, automate routine workflows, enforce SOPs, generate certificates of analysis, and provide audit trails that hold up to regulatory scrutiny.
Top LIMS Software Comparison (2026)
The table below provides an at-a-glance comparison of the most widely used LIMS platforms. This is an independent overview — no vendor has paid for their position in this table.
LIMS Platform | Best For | Deployment | Compliance | Starting Price |
|---|---|---|---|---|
LabWare LIMS | Large enterprise / pharma | Cloud, On-premise, SaaS | GxP, 21 CFR Part 11, ISO 17025 | Custom quote |
LabVantage | Multi-site enterprise | Cloud, On-premise | GxP, 21 CFR Part 11 | Custom quote |
Benchling | R&D / biotech / life science | Cloud (SaaS) | 21 CFR Part 11 | From ~$200/user/mo |
QBench | Testing labs, diagnostics, SMEs | Cloud (SaaS) | CLIA, HIPAA, ISO | Tiered SaaS pricing |
Lockbox LIMS | Clinical & diagnostics labs | Salesforce-native cloud | CLIA, HIPAA, CAP | Custom quote |
SampleManager (Thermo) | Thermo Fisher-heavy labs | Cloud, On-premise | GxP, ISO 17025 | Custom quote |
Clarity LIMS (Veraciti) | Genomics / NGS labs | Cloud, On-premise | ISO 17025 | Custom quote |
CloudLIMS | Small to mid-sized labs | Cloud (SaaS) | ISO 17025, GLP | From ~$300/mo |
STARLIMS (Abbott) | Regulated / pharma / clinical | Cloud, On-premise | GxP, 21 CFR Part 11 | Custom quote |
Scispot | AI-driven biotech / startups | Cloud (SaaS) | 21 CFR Part 11 | Custom / usage-based |
Labguru | Biotech / small pharma R&D | Cloud (SaaS) | 21 CFR Part 11 | From ~$100/user/mo |
LABWORKS | Environmental / industrial / QC | Cloud, On-premise | ISO 17025, GLP | Custom quote |
Independent note: Vendor pricing is rarely published openly. Most enterprise LIMS require a scoping call before any quote is issued. Budget $20,000–$500,000+ for implementation on enterprise systems, and $200–$1,500/user/month for SaaS mid-market platforms. |
Core Features of LIMS Software
Not all LIMS platforms are equal, but the following capabilities define what a modern LIMS should do. Use this list as your baseline checklist when evaluating vendors.
1. Sample Management and Tracking
The foundational capability of any LIMS. A robust sample tracking module eliminates the most common source of lab errors: lost, mislabeled, or untraceable samples.
Sample registration and accessioning
Barcode or RFID labeling
Chain of custody tracking from receipt to disposal
Sample storage location management
Aliquoting and sub-sample tracking
2. Workflow and Test Management
LIMS software should model your lab’s real workflows, not force you to adapt to rigid templates.
Configurable test request and routing
SOP enforcement and protocol templates
Queue management and workload balancing
Instrument scheduling and assignment
3. Result Entry and Data Capture
Manual data entry is the enemy of data integrity. Look for:
Direct instrument interfacing (bi-directional where possible)
Automatic result import and calculation
Out-of-specification (OOS) alerts and flagging
Result review and approval workflows
4. Reporting and Certificates of Analysis (CoA)
Configurable report templates (including CoA)
Automated report generation and distribution
Real-time dashboards and KPI tracking
Export in standard formats (PDF, CSV, HL7, FHIR for clinical)
5. Audit Trails and Data Integrity
In regulated labs, this is non-negotiable.
Full electronic audit trail (who did what, when, and why)
Controlled access and role-based permissions
Electronic signatures (21 CFR Part 11 compliant)
Data archiving and retention policies
6. Integration Capabilities
Laboratory instruments and analyzers (via ASTM, HL7, OPC-UA)
ERP systems (SAP, Oracle)
ELN software
Clinical systems (HIS/LIS/EHR) for healthcare labs
Customer portals for external sample submission
7. Inventory and Reagent Management
Reagent and consumable stock tracking
Lot number and expiry management
Automatic reorder alerts
Cold storage and chain-of-custody documentation
LIMS vs ELN vs SDMS: What’s the Difference?
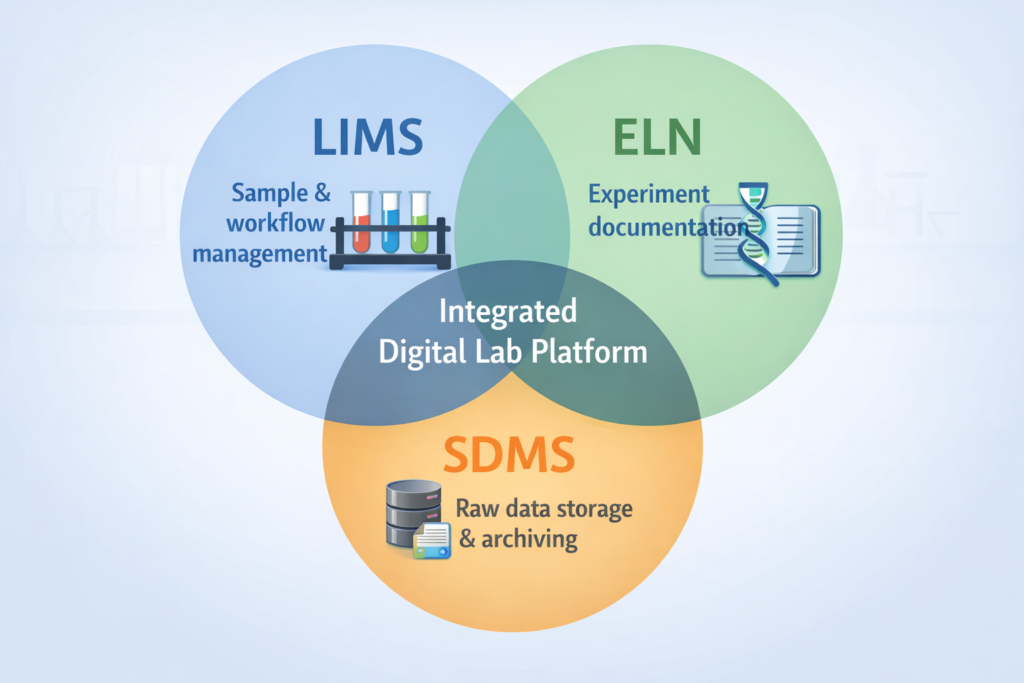
One of the most common sources of confusion when scoping a lab informatics project is understanding how LIMS, ELN, and SDMS differ — and when you need more than one.
LIMS is operational. It manages samples, workflows, quality, and compliance. It answers the question: “What happened to this sample and is the result valid?”
ELN (Electronic Lab Notebook) is research-oriented. It captures experiments, hypotheses, methods, and observations. It answers: “What did we do and why?”
SDMS (Scientific Data Management System) is archival. It stores raw instrument data files. It answers: “Where is the original data file and is it intact?”
Dimension | LIMS | ELN | SDMS |
|---|---|---|---|
Primary focus | Sample & workflow management | Experiment documentation | Raw data storage & archiving |
Main users | Lab technicians, QC managers | Researchers, scientists | Data managers, IT |
Regulatory role | System of record for operations | IP protection | Data integrity & archiving |
Common in | QC labs, clinical, pharma mfg | R&D, academic, early discovery | Analytical labs, pharma R&D |
Many modern platforms blur these boundaries. Benchling combines LIMS and ELN in a single R&D platform. LabWare and LabVantage offer ELN add-ons. The trend is toward unified Digital Lab Platforms that integrate all three — but whether you need an integrated suite or separate best-of-breed tools depends heavily on your context.
LIMS Software by Industry
LIMS requirements vary significantly by industry. A pharmaceutical QC lab faces entirely different compliance demands than an environmental testing lab or a genomics research facility.
Pharmaceutical and Biotech LIMS
Pharma and biotech labs operate under some of the most demanding regulatory frameworks in the world. Your LIMS must support:
21 CFR Part 11 compliance (FDA electronic records and signatures)
GxP alignment (GMP, GLP, GCP) with full validation documentation
ALCOA+ data integrity principles (Attributable, Legible, Contemporaneous, Original, Accurate)
Stability testing and storage condition management
Full IQ/OQ/PQ validation packages from the vendor
Top LIMS for pharma: LabWare, LabVantage, STARLIMS, SampleManager (Thermo), Benchling (R&D phase)
Clinical and Diagnostic Laboratory LIMS
CLIA compliance (Clinical Laboratory Improvement Amendments, US)
CAP accreditation support
HIPAA-compliant data handling
HL7 and FHIR integration with EHR/HIS systems
Reflex testing and autoverification rules
Top LIMS for clinical: Lockbox LIMS, QBench, Orchard Software, SCC Soft Computer
Environmental and Water Testing LIMS
Chain of custody documentation from field collection to result
Regulatory method management (EPA 200.7, SM 2540C, etc.)
Turnaround time tracking and client portal access
Multi-matrix sample management (water, soil, air, sediment)
Top LIMS for environmental: LABWORKS, LabWare, SampleManager
Food and Beverage Testing LIMS
Specification management for raw materials and finished products
Supplier qualification and COA management
Out-of-spec investigation workflows
Top LIMS for food & beverage: LabWare, QBench, CloudLIMS
Genomics and NGS Laboratories
Integration with sequencing instruments
Sample and library preparation tracking
FASTQ file management and pipeline triggering
High throughput plate-based workflows
Top LIMS for NGS: Clarity LIMS (Veraciti), Benchling, GenoLogics
Academic and Research Laboratories
Lightweight onboarding with minimal IT
Flexible sample and inventory tracking
ELN integration or combined ELN/LIMS
Multi-group or multi-PI access management
Top LIMS for academic research: Benchling, Labguru, CloudLIMS, Scispot
How to Choose a LIMS: The Lab Manager’s Buying Guide
Choosing a LIMS is a long-term commitment. Enterprise implementations typically last 10–20 years. Getting the decision right requires more than reading vendor marketing materials. If you’re interested in knowing more read our atricle about lims common implementation challenges.
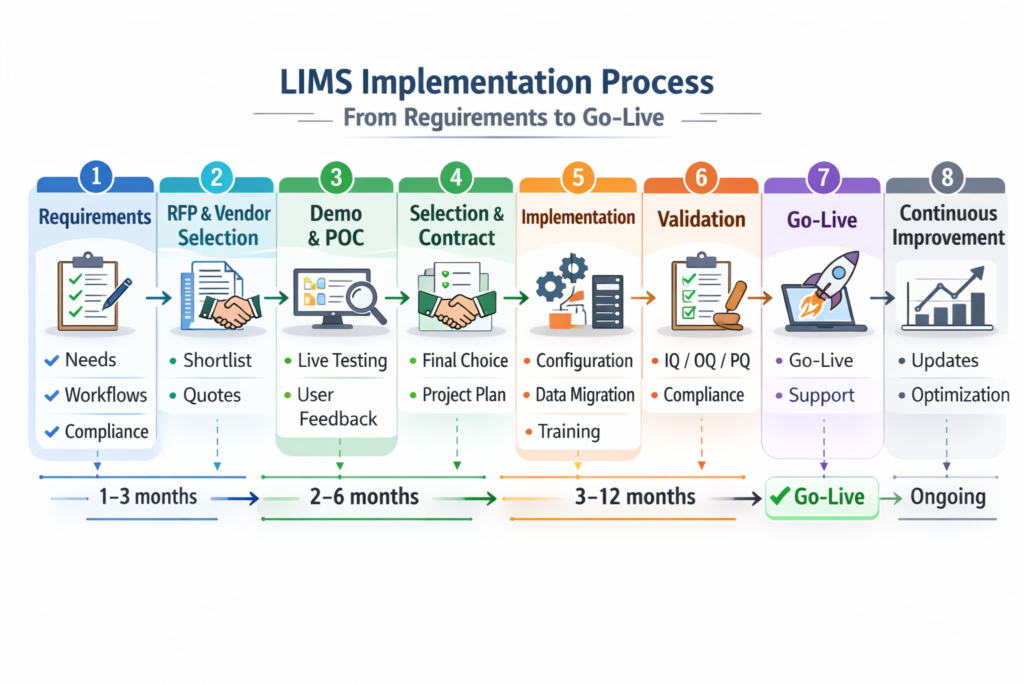
Step 1: Define Your Requirements Before Talking to Vendors
The single biggest mistake labs make is starting with vendor demos before documenting their own requirements. Before any outreach, document:
Current pain points: What is breaking with your existing system or spreadsheets?
Regulatory obligations: Which regulations and standards apply to your lab?
Sample volume and complexity: How many samples per day/week? How many test types?
Integration needs: Which instruments and enterprise systems must the LIMS connect with?
User base: How many users, across how many sites?
Deployment preference: Is cloud/SaaS acceptable, or do you need on-premise?
Step 2: Separate Must-Haves from Nice-to-Haves
Create a weighted requirements matrix. Give higher scores to capabilities that directly impact compliance, data integrity, or patient/product safety. Design features and UI niceties should be weighted lower.
Step 3: Evaluate Total Cost of Ownership (TCO)
The license fee is often the smallest part of the cost. Factor in:
Implementation and configuration fees (often 2–5x the annual license cost for enterprise systems)
Validation costs (IQ/OQ/PQ for regulated labs)
Training costs (initial and ongoing)
Annual maintenance and support fees (typically 18–22% of license value)
Internal IT resources required for maintenance
Step 4: Assess Vendor Stability and Roadmap
How long has the vendor been in business?
Is the company independently owned, PE-backed, or part of a large corporation?
What does the product roadmap look like? Are they investing in AI, cloud, and integrations?
What is the customer retention rate?
Step 5: Insist on a Realistic POC (Proof of Concept)
Any credible LIMS vendor should be willing to demonstrate their system against your actual use cases, not generic demos. Ask them to configure a representative workflow from your lab and walk through it with your team.
Step 6: Talk to Existing Customers
References provided by the vendor will always be positive. Go further: look for user reviews on G2, Capterra, or industry forums. Ask in LinkedIn groups or professional associations for unbiased feedback.
LIMS Compliance and Validation
For labs in regulated industries, LIMS compliance is not optional. Here’s an overview of the most important frameworks. We explore this matter in our lims validation article.
21 CFR Part 11 (FDA, USA)
Governs electronic records and electronic signatures in FDA-regulated environments. A compliant LIMS must provide audit trails, access controls, and validated electronic signatures. This applies to pharmaceutical, medical device, and food manufacturing labs under FDA oversight. Look here if you want to find 21 CFR Part 11 Guide from the FDA.
EU Annex 11 (EMA, Europe)
The European equivalent to 21 CFR Part 11 for computerized systems in GMP environments. EU Annex 11 applies to pharma labs operating under EMA oversight.
ISO 17025
The international standard for testing and calibration laboratories. Accredited labs must demonstrate that their measurement processes are valid and traceable. LIMS systems supporting ISO 17025 need robust method management, uncertainty tracking, and proficiency testing support.
CLIA and CAP (Clinical Labs, USA)
Clinical labs must comply with CLIA regulations governing testing quality for human diagnostic purposes. CAP-accredited labs have additional inspection requirements that the LIMS must support through documentation and audit trails.
GAMP 5 and CSV (Computer System Validation)
Most enterprise LIMS in regulated pharma environments must be validated per the GAMP 5 framework (Good Automated Manufacturing Practice). Vendors typically provide an IQ/OQ/PQ validation package. Always verify this is included and maintained with software updates.
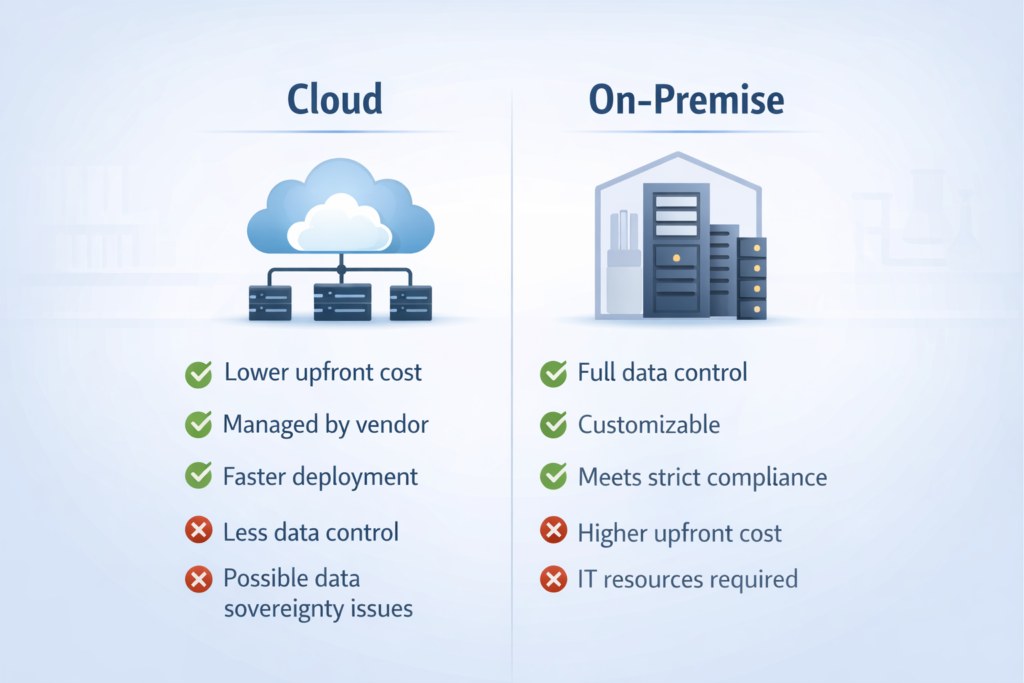
Cloud LIMS vs On-Premise LIMS: Which Is Right for You?
Cloud / SaaS LIMS
Lower upfront cost; subscription-based pricing
Vendor manages infrastructure, updates, and security
Faster implementation timelines (weeks vs months)
May raise data sovereignty concerns in certain regulated environments
Best for: growing labs, SMEs, multi-site organizations, labs without dedicated IT
On-Premise LIMS
Higher upfront investment; full control over infrastructure
Requires in-house IT for maintenance, backup, and updates
Preferred by labs with strict data residency requirements
Better for: large enterprises with IT resources, labs under strict data sovereignty rules
Private Cloud / Hybrid
A middle ground increasingly offered by major vendors. A dedicated cloud instance gives the convenience of SaaS with more control over data. Higher cost than standard SaaS but lower than full on-premise.
The market has shifted decisively toward cloud deployment. Most new LIMS implementations in 2025–2026 are cloud-first, even in heavily regulated environments. The major enterprise vendors (LabWare, LabVantage, STARLIMS) all now offer validated SaaS options.
LIMS Software Pricing: What to Expect
LIMS pricing is notoriously opaque. Here’s a realistic breakdown of what different categories of solution typically cost. If you want to learn more about how lims pricing models work, we have dedicated a full article to this subject.
Category | Price Range | Examples | Implementation |
|---|---|---|---|
SaaS / SME | $150–$1,500/user/mo | QBench, CloudLIMS, Labguru | Days to weeks |
Mid-Market | $20K–$150K license | Scispot, Lockbox LIMS, LABWORKS | 2–6 months |
Enterprise | $100K–$1M+ total | LabWare, LabVantage, STARLIMS | 6–24 months |
For a detailed breakdown of LIMS pricing models — including perpetual vs subscription, modular pricing, and hidden costs — see our article: LIMS Software Pricing Models Explained on labsoftwareguide.com |
Top LIMS Vendors: Independent Reviews
LabWare LIMS

LabWare is widely considered the gold standard for enterprise pharmaceutical and industrial LIMS, with deployments in over 10,000 laboratories across 125 countries. Its breadth of functionality is unmatched — lot management, stability testing, environmental monitoring, instrument interfacing, and configurable dashboards all ship out of the box.
The flip side: LabWare is complex. Implementation projects routinely take 12–18 months and require significant internal resources or a specialist partner. For labs without dedicated LIMS administrators, it can feel like piloting a 747 when you needed a propeller plane.
Best for: Large pharma, industrial QC, multi-site enterprise labs with internal IT resources.
LabVantage

LabVantage competes squarely with LabWare at the enterprise tier. Its modular architecture allows labs to configure industry-specific solutions — biobanking, food and beverage, contract testing, public health — on top of a common core platform. Its integrated ELN, LES, and SDMS components make it attractive for organizations looking to consolidate multiple lab informatics tools.
Best for: Enterprise organizations with complex, multi-departmental lab informatics needs.
Benchling

Benchling has become the dominant LIMS choice for life science R&D and biotech startups. Its modern interface, molecular biology tooling (sequence editor, plasmid maps, chemical registration), and native ELN integration make it a natural choice for discovery-phase scientists who want a single platform for their entire research workflow.
Best for: Biotech and pharma R&D labs, CROs, startups scaling from early discovery to IND-enabling studies.
QBench

QBench has carved out a strong position in the testing, diagnostics, and analytical lab segment. Its cloud-native architecture, fast implementation timelines, and developer-friendly API have made it popular among testing labs that need to get operational quickly. G2 users consistently rate it #1 for ease of use among LIMS platforms.
Best for: Commercial testing labs, diagnostics, cannabis testing, environmental labs seeking fast time-to-value.
STARLIMS (Abbott)

STARLIMS is a compliance-first enterprise LIMS with a strong track record in pharmaceutical QC and clinical laboratory environments. Its mobile capabilities and cloud-friendly architecture are strengths. Implementation is often routed through a partner network rather than the vendor directly.
Best for: Pharmaceutical QC, clinical labs, and regulated environments with established IT teams.
Scispot

Scispot combines traditional sample and workflow management with AI-driven automation. Its code-first approach to customization gives technically sophisticated labs significant flexibility — but labs without developer resources won’t unlock its full potential.
Best for: AI-forward biotech startups and labs with strong in-house technical teams.
CloudLIMS

CloudLIMS is a solid, affordable option for smaller labs that need core LIMS functionality without the complexity or cost of enterprise platforms. The system covers sample management, inventory, instrument management, and ISO 17025-supporting features at an accessible price point.
Best for: Small to mid-sized labs with straightforward workflows and budget constraints.
LIMS vs Spreadsheets: When to Make the Switch
Despite all the sophistication of modern LIMS platforms, a surprising number of labs still run their sample tracking on Excel. The case for transitioning is compelling:
Data errors: Spreadsheets have no built-in controls to prevent incorrect entries, formula overwrites, or accidental deletions.
Auditability: You cannot maintain a defensible, immutable audit trail in Excel.
Scalability: Most labs hit their limit somewhere between 50 and 200 samples per day.
Compliance risk: Any FDA, ISO, or CLIA auditor who finds a spreadsheet managing quality-critical data will ask uncomfortable questions.
The typical trigger points for LIMS adoption are: a failed audit, a significant quality event traced back to a data error, a major increase in sample volume, or a new regulatory requirement.
Frequently Asked Questions About LIMS Software
What does LIMS stand for?
LIMS stands for Laboratory Information Management System. It is software designed to manage laboratory samples, workflows, data, and compliance processes.
What is the difference between LIMS and LIS?
LIMS is a broad term used across industries including pharma, environmental, food, and industrial labs. LIS (Laboratory Information System) typically refers to clinical/hospital laboratory systems. Functionally, they overlap significantly, but LIS systems have stronger healthcare integration (HL7, EHR) while LIMS systems tend to have deeper sample and workflow management for non-clinical environments.
How long does it take to implement a LIMS?
It depends on the system and scope. SaaS LIMS for SMEs can be deployed in days to weeks. Mid-market systems typically take 2–6 months. Full enterprise LIMS implementations (LabWare, LabVantage) routinely take 12–24 months when validation is included.
Do I need to validate my LIMS?
If you operate in a regulated environment under FDA (21 CFR Part 11), EU GMP (Annex 11), or similar frameworks, yes — your LIMS must be validated as a computerized system. This typically involves IQ/OQ/PQ protocols and vendor-supplied validation documentation. ISO 17025 accredited labs should also verify that their LIMS supports their quality system requirements.
What is the difference between an open-source LIMS and a commercial LIMS?
Open-source LIMS platforms (such as Bika LIMS / Senaite) offer the source code freely, reducing licensing costs but increasing the burden on your internal team to configure, maintain, and validate. Commercial LIMS vendors provide support, validation packages, and implementation services. For regulated labs, the validation support and SLAs of commercial vendors are usually worth the investment.
Can a LIMS integrate with my laboratory instruments?
Yes. Most modern LIMS platforms support bidirectional instrument integration via ASTM E1381, ASTM E1394, RS-232, or modern IP-based protocols. Middleware solutions (like Data Innovations or Roper) can bridge legacy instruments. Always confirm which specific instruments are on the vendor’s validated integration list before purchase.
What is a cloud LIMS?
A cloud LIMS is a Laboratory Information Management System hosted on cloud infrastructure (AWS, Azure, Google Cloud) and accessed via a web browser, rather than installed on local servers. Cloud LIMS typically offer lower upfront costs, automatic updates, and greater accessibility for multi-site or remote teams.
Conclusion: Finding the Right LIMS for Your Laboratory
There is no universally ‘best’ LIMS — the right system depends on your laboratory type, regulatory environment, team size, budget, and long-term growth plans.
For large pharma or industrial QC operations, LabWare and LabVantage remain the benchmark. For R&D-focused biotech labs, Benchling is hard to beat. For mid-sized testing and diagnostics labs that want fast deployment, QBench or CloudLIMS offer compelling value.
The most important advice: define your requirements before you talk to vendors. Get input from the bench-level scientists who will use the system daily. Run a realistic proof of concept. Check total cost of ownership, not just the license fee.
This guide is updated regularly as the market evolves. Bookmark labsoftwareguide.com and return as your evaluation progresses. |
Explore More LIMS Resources on LabSoftwareGuide
LIMS Software Pricing Models Explained
Common LIMS Implementation Challenges
LIMS vs ELN: Which Does Your Lab Need?
Open-Source LIMS: Pros, Cons and When It Makes Sense
How to Write a LIMS Requirements Document
Best LIMS for Small Laboratories
21 CFR Part 11 Compliance: What Your LIMS Must Do
LabSoftwareGuide is an independent editorial resource. We do not accept payment for vendor rankings or editorial positions. Some articles may contain clearly disclosed sponsored content or affiliate links.
Interested in getting in touch with our LIMS partners?
About Lab Software Guide Editorial Team
Who are we ?
The LabSoftwareGuide editorial team is led by a laboratory informatics specialist with extensive experience in the commercial side of lab software — including LIMS, ELN, and bioinformatics platforms across pharma, biotech, and life science environments.
Our editorial approach is built on that experience: we understand how lab software is evaluated, purchased, and implemented, and we know the questions that lab managers, scientists, and IT teams actually struggle with when choosing between platforms.
How we work ?
Every guide and review on LabSoftwareGuide is produced through a combination of hands-on platform testing, vendor interviews, and analysis of real user feedback from G2, Capterra, and laboratory community forums. We cross-reference commercial claims against documented user experience to give you assessments that reflect reality — not marketing materials.
Our Editorial Independence
LabSoftwareGuide accepts sponsored content and commercial partnerships with software vendors. These are always clearly labeled. Our editorial guides, pillar pages, and independent reviews are produced separately from any commercial relationship and are not influenced by vendor partnerships.
Our focus: We cover laboratory information management systems (LIMS), electronic lab notebooks (ELN), and related lab software for professionals working in pharma, biotech, clinical, environmental, and research laboratories.

Open-Source ELN and LIMS: The Honest Guide for 2026
The promise of open-source laboratory software is compelling: no licence fees, full access to the source code, unlimited users, and the freedom to customise every workflow. In a market where

ISO 17025 and LIMS: What Your Software Must Support
For testing and calibration laboratories seeking or maintaining accreditation, ISO/IEC 17025:2017 is the definitive benchmark. Published by the International Organization for Standardization, it sets out the requirements for competence, impartiality,

How to Choose a LIMS: 10 Questions to Ask Before Buying
A practical buyer’s guide for lab managers, quality directors, and IT teams evaluating Laboratory Information Management Systems. Selecting a Laboratory Information Management System (LIMS) is one of the most consequential