In regulated laboratory environments, few topics are as critical—and as misunderstood—as LIMS Validation Explained. Whether you operate in pharmaceutical manufacturing, clinical diagnostics, biotech R&D, or environmental testing, validating your Laboratory Information Management System (LIMS) is not optional. It is a regulatory and operational necessity.
Understanding LIMS Validation Explained is essential for lab directors, QA managers, IT teams, and compliance officers. A properly validated LIMS protects data integrity, ensures regulatory compliance, and safeguards your organization against costly audit findings.
Let’s break it down clearly and strategically.
Introduction to LIMS Validation Explained
A LIMS manages sample tracking, workflows, reporting, and data storage. When that system operates in a regulated environment, authorities require proof that it functions correctly and consistently.
Validation provides that proof.
Agencies such as the U.S. Food and Drug Administration expect computerized systems that impact product quality or patient safety to be validated. Without proper validation documentation, a laboratory risks warning letters, fines, or operational shutdown.
Simply put: validation protects both your data and your reputation.
What Is LIMS Validation?
Definition and Purpose
LIMS validation is a documented process that demonstrates a LIMS system performs as intended, consistently and reliably, according to predefined requirements.
It answers three fundamental questions:
- Does the system meet user requirements?
- Does it function as designed?
- Does it maintain data integrity and compliance?
Validation ensures traceability from business needs to system functionality.
Why Validation Is Mandatory in Regulated Labs
In regulated sectors, digital records are treated with the same seriousness as physical records. Any system that stores, modifies, or transmits regulated data must prove:
- Accuracy
- Reliability
- Consistency
- Security
Without validation, there is no formal assurance that the system works as required.
Regulatory Frameworks Governing LIMS Validation

21 CFR Part 11
One of the most recognized regulations is 21 CFR Part 11. It governs electronic records and electronic signatures.
It requires:
- Secure user authentication
- Audit trails
- System validation
- Record integrity controls
Compliance is mandatory for companies regulated by the U.S. Food and Drug Administration.
GxP, GMP, and GLP Requirements
Good Practice (GxP) guidelines apply to:
- GMP (Good Manufacturing Practice)
- GLP (Good Laboratory Practice)
- GCP (Good Clinical Practice)
All require validated computerized systems that affect product quality or data reliability.
ISO Standards and Data Integrity Guidelines
Standards such as ISO 17025 and ISO 9001 emphasize:
- Traceability
- Controlled documentation
- Risk management
- Continuous improvement
Validation supports all these requirements.
Why LIMS Validation Is Critical for Modern Laboratories
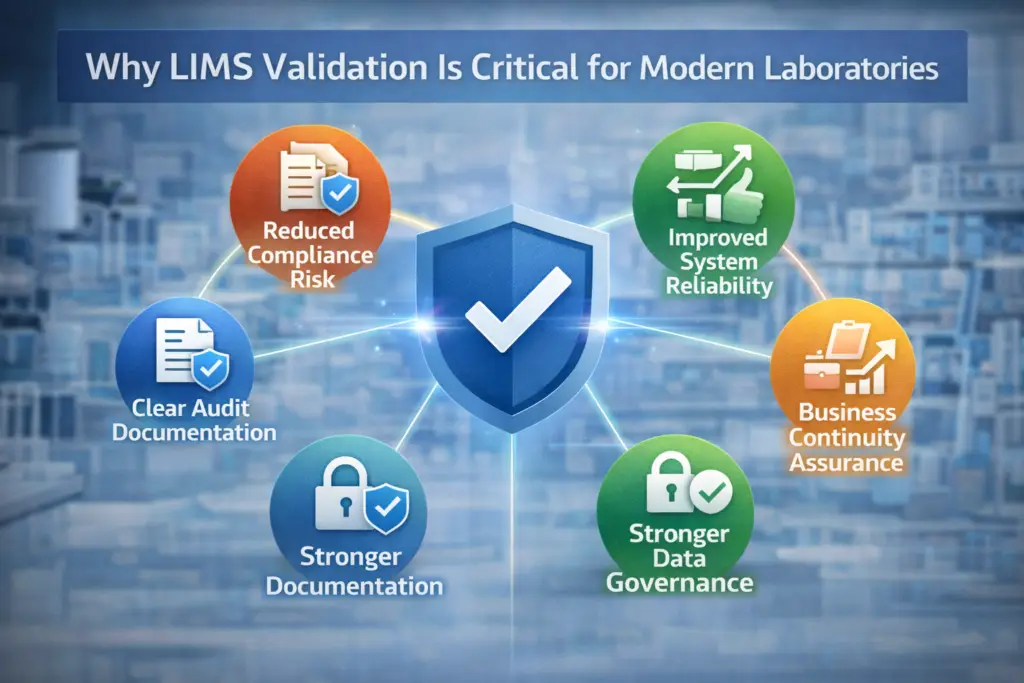
Validation is not just regulatory bureaucracy.
It provides:
- Reduced compliance risk
- Improved system reliability
- Clear documentation for audits
- Stronger data governance
- Business continuity assurance
Without validation, laboratories face operational uncertainty and reputational risk.
In short, validation builds trust.
The Risk-Based Approach to LIMS Validation
Modern validation strategies follow a risk-based methodology.
Instead of validating everything equally, laboratories assess:
- What data impacts product quality?
- What processes affect patient safety?
- What system functions carry the highest risk?
High-risk components receive deeper testing and documentation.
This approach aligns with guidance from international regulators and ensures efficient use of validation resources.
The LIMS Validation Lifecycle Explained
The validation lifecycle typically follows structured phases.
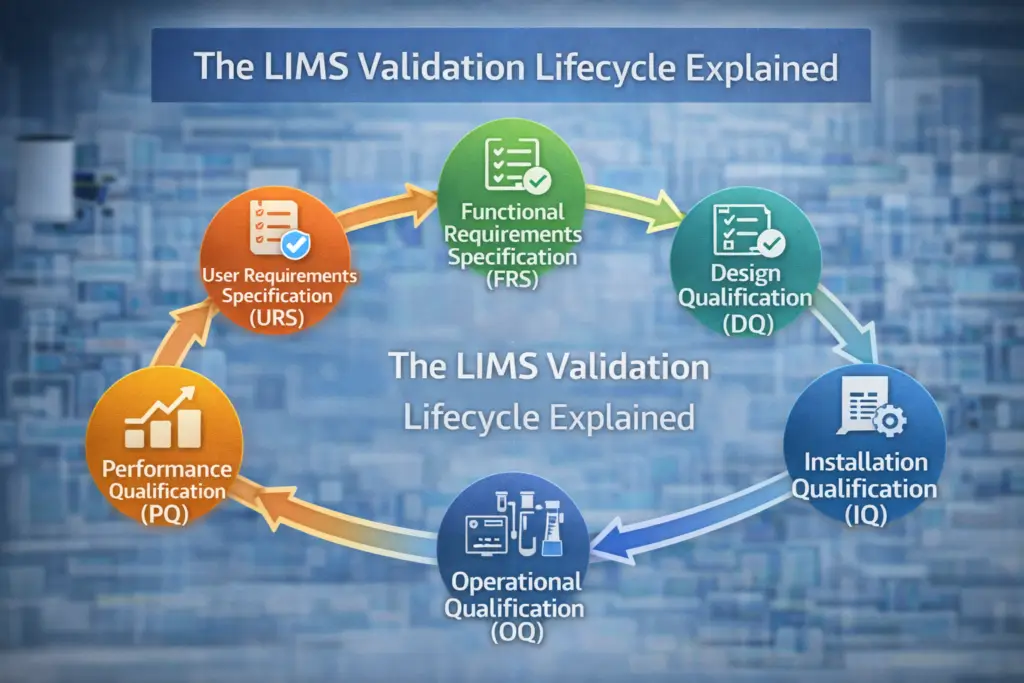
User Requirements Specification (URS)
The URS defines what the lab needs from the LIMS.
It includes:
- Sample management needs
- Reporting requirements
- Compliance expectations
- Integration requirements
The URS becomes the foundation of validation.
Functional Requirements Specification (FRS)
The FRS describes how the system will meet those needs.
It translates business needs into technical functions.
Design Qualification (DQ)
DQ confirms that the selected LIMS solution is suitable for its intended purpose.
Installation Qualification (IQ)
IQ verifies that:
- The system is installed correctly
- Hardware and software match specifications
- Environment setup is compliant
Operational Qualification (OQ)
OQ tests that the system operates according to specifications under controlled conditions.
Performance Qualification (PQ)
PQ confirms the system performs effectively in real-world operational conditions.
Together, IQ, OQ, and PQ provide structured evidence of system reliability.
Validation Documentation: What You Must Have
A complete validation package includes:
- Validation Plan
- URS and FRS
- Risk Assessment
- Test Scripts and Protocols
- Test Results
- Traceability Matrix
- Validation Summary Report
During audits, inspectors will request this documentation.
Incomplete records often trigger observations.
Common Mistakes in LIMS Validation
Organizations often make critical errors, including:
- Treating validation as a one-time project
- Poorly defined requirements
- Lack of risk assessment
- Incomplete documentation
- Insufficient change control
- Over-reliance on vendor documentation
Remember: vendors support validation—but ultimate responsibility remains with the laboratory.
Cloud-Based LIMS Validation Considerations
Cloud LIMS solutions introduce additional considerations:
- Infrastructure control
- Data hosting location
- Cybersecurity
- Vendor qualification
- Service Level Agreements (SLAs)
Cloud does not remove validation requirements.
It shifts focus toward vendor audits and shared responsibility models.
Maintaining Validation: Change Control and Periodic Review
Validation does not end at go-live.
Systems evolve through:
- Software updates
- Configuration changes
- New integrations
- Regulatory updates
Each change must be evaluated under formal change control procedures.
Periodic review ensures the system remains compliant over time.
Ongoing validation maintenance protects long-term integrity.
Frequently Asked Questions
1. Is LIMS validation required for all laboratories?
It is mandatory for regulated laboratories operating under FDA, GMP, GLP, or ISO requirements.
2. How long does LIMS validation take?
Typically several months, depending on complexity and customization.
3. Can vendors provide validation documents?
Yes, but laboratories remain responsible for final validation.
4. Is cloud LIMS easier to validate?
It can simplify infrastructure validation but still requires documentation and risk assessment.
5. What happens during an audit?
Inspectors review validation documentation, change control records, and audit trails.
6. What is the biggest risk of not validating?
Regulatory penalties, warning letters, or operational shutdown.
Conclusion: Why LIMS Validation Is a Strategic Investment
When we talk about LIMS Validation Explained, we are not just discussing compliance checkboxes.
We are discussing:
- Data integrity
- Operational stability
- Regulatory confidence
- Business continuity
Validation transforms a LIMS from a software tool into a trusted compliance asset.
For laboratories operating in regulated industries, LIMS validation is not simply a requirement—it is a strategic safeguard.
Organizations that invest in structured validation reduce risk, strengthen governance, and build long-term operational resilience.
And in today’s highly regulated scientific landscape, that competitive advantage is priceless.



